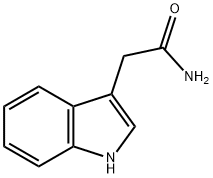
3-Indoleacetamide: Conformational Analysis & Auxin Biosynthesis in Plants
3-Indoleacetamide is a member of the class of indoles that is acetamide substituted by a 1H-indol-3-yl group at position 2. It is an intermediate in the production of plant hormone indole acetic acid (IAA). It has a role as a fungal metabolite, a bacterial metabolite and a plant metabolite. This compound belongs to the class of organic compounds known as 3-alkylindoles. These are compounds containing an indole moiety that carries an alkyl chain at the 3-position.

Conformational Analysis of 3-Indoleacetamide
Tryptophan stands as one of nature's most versatile amino acid precursors, serving as the biosynthetic foundation for an extraordinary array of bioactive molecules with diverse and specialized physiological functions. This aromatic amino acid undergoes complex metabolic transformations that give rise to compounds ranging from essential neurotransmitters to critical plant growth regulators. Among the most significant metabolic pathways are those leading to serotonin (5-hydroxytryptamine), a pivotal neurotransmitter in the central nervous system, and 3-indoleacetic acid (IAA), the primary auxin responsible for plant growth and development. One of the biosynthetic pathways from tryptophan to IAA involves 3-indoleacetamide (IAM) serving as a crucial metabolic intermediate in one of the major routes. This pathway represents a fascinating example of how nature has evolved distinct molecular architectures to achieve specific biological outcomes, despite starting from a common precursor. In this context, we have performed the first rotational spectroscopic study of 3-Indoleacetamide using LA-CP-FTMW and LA-MB-FTMW spectroscopies to identify the most relevant conformers of this crucial metabolic intermediate. These precise structural results will be compared with those of tryptophan, tryptamine, serotonin and IAA.[1]
To identify the most energetically favorable geometries of neutral 3-Indoleacetamide, we performed an extensive conformational analysis using molecular mechanics approaches. This search yielded 12 distinct structures spanning an energy window of 2500 cm−1 (30 kJ/mol). Each structure underwent geometry optimization via quantum mechanical methods implemented in the Gaussian program suite, following the computational protocol detailed in the methodology. All 12 initial conformers ultimately relaxed to a single, lowest-energy structure. The nuclear coupling constants are similar but still slightly different from those of pyrrole and indole, with values falling between those of both systems. This demonstrates how small variations close to the nitrogen atom affect the quadrupole coupling constants. Comparing the quadrupole coupling constants of 3-Indoleacetamide with those of tryptamine confirms that the NaH•••Nr interaction is similar in both systems due to the very similar values observed for the pyrrolic nitrogen. On the other hand, the values for the amino nitrogen are drastically different due to the distinct chemical environments of the nitrogen in -CH2-NH2 for tryptamine and in -C=O-NH2 for 3-Indoleacetamide. In the next section, we provide further comparison of the latter through Non-Covalent Interaction (NCI) analysis. This reveals unprecedented structural rigidity within the tryptophan-derived bioactive molecule family, as it represents a dramatic departure from the conformational flexibility characteristic of related neurotransmitters and plant hormones. The acetamide functional group imposes geometric constraints through restricted rotation due to partial double-bond character and the dual hydrogen bonding involving both N-H groups.
The auxin effects of 3-Indoleacetamide in Arabidopsis
IAM was the first definitively identified intermediate used in Trp-dependent auxin biosynthesis pathways in bacteria. Plant pathogens such as agrobacterium and pseudomonas synthesize auxin from Trp when they infect plants. The bacteria-produced auxin alters the growth and developmental patterns of the infected plant cells so that the pathogens can use the plant cells to produce carbon- and nitrogen- rich compounds for their growth. The pathogens convert Trp into 3-Indoleacetamide using the bacterial iaaM Trp-2-monooxygenase and subsequently the pathogen-encoded hydrolase iaaH converts IAM to IAA. Arabidopsis and other plants produce IAM in the absence of a bacterial infection, suggesting that plants may use IAM as an auxin biosynthetic intermediate as well. Furthermore, IAM was proposed as an intermediate in a route that converts IAOx into IAA. It is well known that Arabidopsis and other plants have the capacity to convert 3-Indoleacetamide into IAA. Overexpression of iaaM in Arabidopsis, petunia, and tobacco led to auxin overproduction phenotypes. It is hypothesized that plant hydrolases can convert IAM produced by the iaaM transgene to generate IAA. In this study, we present the identification of two homologous genes that encode 3-Indoleacetamide hydrolases (IAMHs) responsible for converting IAM to IAA in Arabidopsis. Arabidopsis plants that lacked the IAMH activities were resistant to exogenous IAM. Mutations in the IAMH genes suppressed the auxin overproduction phenotypes caused by overexpression of iaaM, the bacterial auxin biosynthetic gene.[2]
Although IAM was the first confirmed Trp metabolite that could serve as an auxin precursor, its role in auxin biosynthesis in plants has never been clarified. In this paper, we uncovered two IAMH genes that encode hydrolases that are capable of converting 3-Indoleacetamide into IAA in Arabidopsis. We showed that Arabidopsis plants lacked the two IAMH genes were resistant to IAM treatments. An obvious candidate is the Amidase1, which has been studied extensively for almost two decades. It will be interesting to further knock out the amidase family of genes in the iamh1 iamh2 double mutant background. Analysis of the higher order mutants in terms of 3-Indoleacetamide sensitivity and developmental phenotypes will provide a more definitive answer to the roles of IAM in auxin biosynthesis and plant development. In summary, we have identified two IAM hydrolase genes that are responsible for converting IAM into IAA in Arabidopsis. We showed that the IAMH genes are required for the auxin overproduction phenotypes caused by 3-Indoleacetamide treatments or by overexpressing the bacterial iaaM gene. Our findings provide a foundation for determining the roles of IAM in auxin biosynthesis and in plant development. Moreover, our findings will also help resolve the molecular mechanisms by which IAOx is converted into IAA.
References
[1]Municio S, Mato S, Alonso JL, Alonso ER, León I. Conformational Analysis of 3-Indoleacetamide: Unveiling Structural Rigidity in the Tryptophan-Derived Bioactive Molecule Family. Molecules. 2025 Oct 22;30(21):4156. doi: 10.3390/molecules30214156. PMID: 41226120; PMCID: PMC12609830.
[2]Gao Y, Dai X, Aoi Y, Takebayashi Y, Yang L, Guo X, Zeng Q, Yu H, Kasahara H, Zhao Y. Two homologous INDOLE-3-ACETAMIDE (IAM) HYDROLASE genes are required for the auxin effects of IAM in Arabidopsis. J Genet Genomics. 2020 Mar 20;47(3):157-165. doi: 10.1016/j.jgg.2020.02.009. Epub 2020 Mar 19. PMID: 32327358; PMCID: PMC7231657.
Lastest Price from 3-Indoleacetamide manufacturers

US $10.00/KG2025-04-21
- CAS:
- 879-37-8
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $10.00/kg2025-04-21
- CAS:
- 879-37-8
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20 ton